Pipeline
We are focused on the discovery and development of potentially best-in-class or first-in-class small molecule therapeutics. To achieve this, we aim to address issues such as tolerability and combinability, resistance, and disease escape through brain metastases.
Our lead product candidates include:

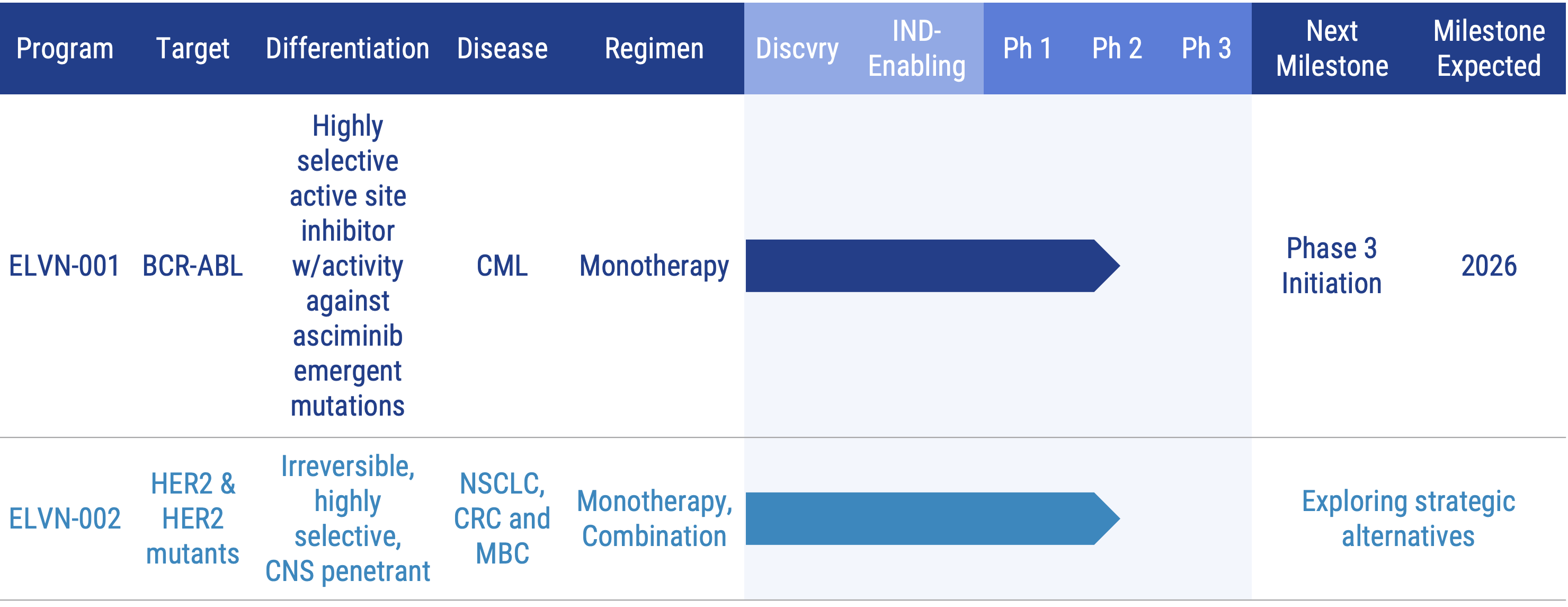


BCR-ABL Program: ELVN-001
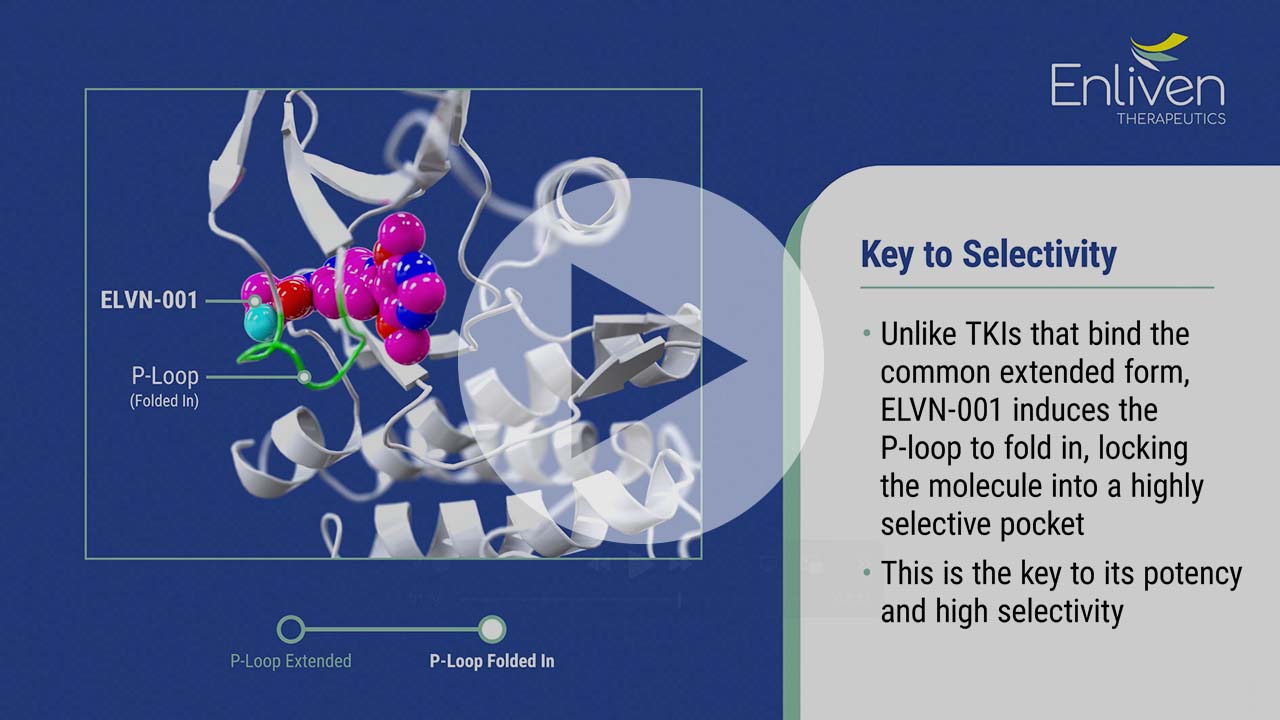
ELVN-001 is a potent, highly selective, small molecule kinase inhibitor designed to specifically target the breakpoint cluster region – Abelson (“BCR-ABL”) gene fusion, the oncogenic driver for patients with chronic myeloid leukemia (“CML”). It targets the ATP-binding site of the ABL1 kinase domain and binds to a unique P-loop “folded-in” active conformation of ABL1, creating a narrow selectivity tunnel.
Although the approval of BCR-ABL tyrosine kinase inhibitors (“TKIs”) changed prognosis of CML from an often fatal disease to a manageable chronic condition, patients still face several barriers that limit their ability to achieve durable responses, maintain long-term treatment success, and sustain a good quality of life. Patients can develop primary or secondary resistance to currently available TKIs, preventing them from achieving or maintaining the level of response required for long-term survival. In addition, the currently available ATP-competitive TKIs (e.g. nilotinib, dasatinib) have off-target activity that may lead to adverse events, potentially requiring dose reductions or treatment discontinuation. All approved TKIs are associated with drug-drug interactions that can affect the safety and efficacy of some medications, and most BCR-ABL TKIs must be taken either with food or on an empty stomach, making adherence more difficult. These factors underscore the significant global opportunity for therapies that can address these unmet needs.
As a highly selective ATP-competitive inhibitor, ELVN-001 is differentiated from other ATP-competitive inhibitors in that it avoids off-target activity that may cause adverse events. With this potential additional safety margin, ELVN-001 may be optimally dosed to drive better efficacy. Additionally, ELVN-001 potentially represents a complementary option to allosteric BCR-ABL inhibitors (e.g., asciminib) due to its distinct mechanism of action and high selectivity which may play an increasingly important role in the standard of care for CML. Specifically, ELVN-001 was designed to have activity against emerging mutations known to confer resistance to allosteric TKIs, including asciminib. ELVN-001 was also designed to be a more attractive option for patients with comorbidities, on concomitant medications or desiring more freedom from stringent administration requirements.
ELVN-001 is currently being evaluated in the Phase 1 ENABLE clinical trial in heavily pretreated patients with CML, and we plan to initiate ENABLE-2, a 2L+ Phase 3 pivotal trial in the second half of 2026. The most recent data is available on the Program Presentation & Publications section of the website.
HER2 Program: ELVN-002
ELVN-002 is being evaluated in a Phase 1 trial as a monotherapy agent in people with HER2+ and HER2 mutant tumors. As part of that trial, we enrolled patients with HER2+ metastatic breast cancer (MBC) in an exploratory cohort in combination with ado-trastuzumab emtansine and patients with HER2+ NSCLC in an exploratory cohort in combination with trastuzumab deruxtecan. To learn more, please visit www.clinicaltrials.gov (NCT05650879).
ELVN-002 is also being evaluated in combination with trastuzumab with or without chemotherapeutic agents in people with HER2+ solid tumors. To learn more about the Phase 1 study, please visit www.clinicaltrials.gov (NCT06328738).
In May 2025, we made the decision to explore strategic alternatives for ELVN-002 so that we could prioritize ELVN-001.
Additional Programs
Our drug development strategy is target and indication agnostic, rooted in validated biology and differentiated chemistry. Our pipeline reflects this approach, featuring medicines enabled by our expertise in complex medicinal chemistry to deliver new and improved treatments for patients.
Program Presentations & Publications
Recently Published
ENABLE: Updated Efficacy and Safety Results of ELVN-001, A Novel Selective ATP-Competitive Inhibitor of BCR::ABL1, in Patients with Previously Treated CP-CML
European Hematology Association Congress (oral presentation) | June 2026
Preliminary safety and efficacy of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with CML driven by atypical fusion transcripts
67th Annual American Society of Hematology Annual Meeting | December 2025
To Improve Upon a Miracle Drug: Overcoming Drug Resistance & Intolerance in CML
Drug Development & Delivery | November 2025
ENABLE: A Phase 1 Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
European Society of Hematology International Chronic Myeloid Leukemia Foundation (ESH-iCMLf) 27th Annual John Goldman Conference | October 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
Society of Hematologic Oncology Annual Meeting (oral presentation) | September 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
Society of Hematologic Oncology Annual Meeting (poster) | September 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated chronic myeloid leukemia
Society of Hematologic Oncology Annual Meeting (abstract) | August 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
European Hematology Association Congress (oral presentation) | June 2025
2025
Preliminary safety and efficacy of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with CML driven by atypical fusion transcripts
67th Annual American Society of Hematology Annual Meeting | December 2025
To Improve Upon a Miracle Drug: Overcoming Drug Resistance & Intolerance in CML
Drug Development & Delivery | November 2025
ENABLE: A Phase 1 Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
European Society of Hematology International Chronic Myeloid Leukemia Foundation (ESH-iCMLf) 27th Annual John Goldman Conference | October 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
Society of Hematologic Oncology Annual Meeting (oral presentation) | September 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
Society of Hematologic Oncology Annual Meeting (poster) | September 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated chronic myeloid leukemia
Society of Hematologic Oncology Annual Meeting (abstract) | August 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
European Hematology Association Congress (oral presentation) | June 2025
ENABLE: A Phase 1a/1b Study of ELVN-001, a selective active site inhibitor of BCR::ABL1, in patients with previously treated CML
European Hematology Association Congress (abstract) | May 2025
Development and application of a mechanistic pharmacokinetic pharmacodynamic (PKPD) model to predict anti-chronic myeloid leukemia (CML) effects of tyrosine kinase inhibitors
American Association for Cancer Research Annual Meeting | April 2025
ELVN-001, a highly selective ATP-competitive ABL1 tyrosine kinase inhibitor for the treatment of chronic myeloid leukemia alone or in combination with asciminib
American Association for Cancer Research Annual Meeting | April 2025
ELVN-002, a potent, selective HER2 inhibitor with a differentiated binding mode conferring the potential for enhanced efficacy in combination with HER2-targeting antibody-drug conjugates
American Association for Cancer Research Annual Meeting | April 2025
ELV-3111, a type 1 pan-RAF inhibitor, that safely combines with MEK inhibitors for enhanced anti-tumor activity in NRAS and BRAF mutant cancers including the most common mechanisms of BRAF inhibitor clinical resistance
American Association for Cancer Research Annual Meeting | April 2025
Mechanism of tumor-selective inhibition of dimeric RAF by a Type 1 RAF inhibitor
American Association for Cancer Research Annual Meeting | April 2025
2024
Application of PKPD Principles to Drive the Discovery and Development of Novel BCR-ABL Tyrosine Kinase Inhibitors
American Conference on Pharmacometrics | November 2024
Preliminary safety and efficacy of ELVN-001, a selective active site inhibitor of BCR::ABL1 in CML
European Society of Hematology International Chronic Myeloid Leukemia Foundation (ESH-iCMLf) 26th Annual John Goldman Conference | September 2024
ELVN-001 Positive Proof of Concept Data from Phase 1 Clinical Trial of ELVN-001 in Chronic Myeloid Leukemia
Company Hosted Event with Key Opinion Leaders | April 2024
2023
Trials in Progress: Phase 1a/b study of ELVN‑002 in solid tumors with HER2 mutations, amplification, or overexpression
World Congress of Lung Cancer | September 2023
Preclinical Activity of ELVN-002: a Potent, Selective, Irreversible and CNS Penetrant HER2 and pan-HER2 Mutant Small-Molecule Inhibitor for the Treatment of HER2-Driven Malignancies
American Association for Cancer Research Annual Meeting | April 2023
2022
ELVN-001, a Next-Generation, ATP-Competitive ABL1 Tyrosine Kinase Inhibitor for the Treatment of Chronic Myeloid Leukemia
64th American Association of Hematology Annual Meeting and Exposition | December 2022
Trial in Progress: First-in-human study of ELVN-001, a highly selective BCR::ABL1 tyrosine kinase inhibitor, in patients with chronic myeloid leukemia who failed previous TKI therapies
24th Annual John Goldman Conference on Chronic Myeloid Leukemia: Biology and Therapy | October 2022,
64th American Association of Hematology Annual Meeting and Exposition | December 2022